|
1/17/2024 0 Comments Atomic mass of carbonIf they are next to each other, multiply them to get the answer, if one is on top of the other, divide the top one by the bottom one to get the answer. The atomic mass of lithium is 6.94, the naturally occurring isotopes are 6Li 6.015121 amu, and 7Li 7.016003 amu. The atomic mass of an element is the average mass of the atoms of an element. To get the formula from the triangle, cover up the value you want to calculate and use the remaining two values. 
To remember the formulas in this section, you can use the formula triangles. For example, about 98.9% of naturally occurring carbon is 12C ('Carbon 12') and about 1.1% is 13C so carbon has an atomic weight of \begin Formula Triangles The atomic weight (or atomic mass) of an element tells us on average how much one atom of a given element weighs, taking into account typical proportions of isotopes. The entire science of organic chemistry, which is the study of carbon compounds, is based on carbon.Contents Toggle Main Menu 1 Atomic Weight 2 Molecular Weight 3 Moles 4 Solutions 4.1 1) Molarity 4.2 2) Mass Concentration 4.3 A note on units for concentrations 4.4 Diluting Solutions 5 Test yourself Atomic Weight No form of life on the planet earth is without carbon in its structure. Isotopic masses can be measured using mass spectroscopy. Every plant, animal, and everything in between that is alive is based on carbon. isotopic mass of carbon-13 atom: 13 nucleons × 1 u/ nucleon 13 u. The most important impact carbon has on the human race is the fact that carbon is the basic building block of life, as we know it. As an example, solid carbon is used to reduce iron from its oxide (Fe+3 to Fe0) in the blast furnace or other similar processes in which metallic elements are reduced from their oxide ores. Its ability to combine with oxygen makes it both a powerful and useful reducing agent (a substance that donates electrons resulting in reduction of the charge of the ion or atom being reduced). Carbon has a particularly strong affinity for oxygen either in the form of gaseous oxygen, or as oxygen contained in chemical combination with other elements. The elements of the periodic table sorted by atomic mass - Electronegativity, 10.811, Boron - Density, 12.0107, Carbon - Melting point, 14.0067, Nitrogen. 
We combine (react) many mers of ethylene. Pure carbon is a relatively reactive element and combines directly with many chemical elements, especially those considered oxidizing agents. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) 28. Depending on the amount of graphite, amorphous carbon, or other contaminating elements, diamond can be found in colors ranging from clear water white through shades of black, gray, yellow, red, orange, blue, and green. In this scale, 1 atomic mass unit (amu) corresponds to 1.66 x 1024 gram. Why 12.01 amu If a sample of carbon was placed in a mass spectrometer the spectrometer would detect two different C atoms, 12 C and 13 C. No single carbon atom has a mass of 12.01 amu, but in a handful of C atoms the average mass of the carbon atoms is 12.01 amu. In a typical sample of carbon-containing material, 98.89 of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. 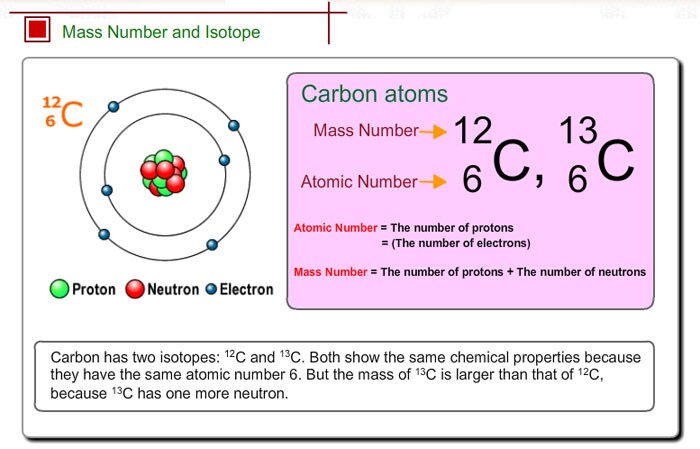
It is expressed as a multiple of one-twelfth the mass of the carbon-12 atom, which is assigned an atomic mass of 12 units. This is the average atomic mass of carbon. The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. Most forms of carbon, excluding diamond, are black to grey-black in color. Atomic mass, the quantity of matter contained in an atom of an element. Carbon-14 (14C): The carbon isotope whose nucleus contains six protons and eight neutrons. Then you have 9893 atoms of 12C and 107 atoms of 13C. Assume that you have, say, 10 000 atoms of carbon. Some of these forms include hexagonal graphite, rhombohedral graphite, diamond, buckminsterfullerene, and amorphous carbon (not really a crystalline form). To find the average atomic mass, you take a certain number of atoms, find the total mass of each isotope, and then divide the total mass of all the atoms by the total number of atoms. There are a number of forms of carbon, known as allotropes, which are composed of pure carbon atoms but are arranged in different crystal lattices. Carbon is one of the few elements that occur in nature in its native or free elemental form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
